Reductive removal of mercury with NaBH 4. For example, the preferred way to produce the Markovnikov alcoholįrom an olefin is to use hydroxymercuration for the addition step followed by Instead methods are used which permit much greater regiochemical control andĪre milder. However, simpleĪcid-catalyzed addition of water is often the least desirable alternative. Thus a common method for the preparation of alcohols. Addition of water across an olefinic dou-ble bond is Aldehydes give primary alcohols while ketonesĪnd olefins are at the same oxidation level and are interconvertible without aĬhange in oxidation level. Milder than LAH and does not require aprotic conditions (an alcohol is often As such it provides a selective way to reduce acidsĪnd produce alcohols in the presence of most other functional groups.Īnd ketones are conveniently reduced by sodium borohydride, which is much Increases the reactivity of the boron hydride and delivers the hydride by an Unique reactivity comes from the fact that borane first forms a Lewis acid –īase complex with the acid and then a boron – carboxylate intermediate which With carboxylic acids than with esters or other acid derivatives. Reduced easily by borane, which is the only reducing agent that reacts faster Carboxylic acids, but not esters, are also Reductive methods have been reported for the preparation of alcohols.Ĭarboxylic acids and esters react vigorously with lithium aluminum hydride Groups and consequently are readily prepared by reduction. Methods for the preparation of alcohols are still among the most usefulĪre at a fairly low oxidation level compared to other oxygen-containing functional The alcohol group, but the alcohol group can be prepared from many other groups and converted to many functional groups. Not only do many important compounds and pharmaceuticals contain Not only do many important compounds and pharmaceuticals contain the alcohol group, but the alcohol group can be prepared from many other groups and converted to many functional groups.Īlcohol functional group is a very important functional group in organicĬhemistry. The alcohol functional group is a very important functional group in organic chemistry. The most reactive site in an alcohol molecule is the hydroxyl group, despite the fact that the O–H bond strength is significantly greater than that of the C–C, C–H and C–O bonds, demonstrating again the difference between thermodynamic and chemical stability.Chapter: Organic Chemistry : Functional Group Synthesis Indeed, the dipolar nature of the O–H bond is such that alcohols are much stronger acids than alkanes (by roughly 10 30 times), and nearly that much stronger than ethers. Consequently, the covalent bonds of this functional group are polarized so that oxygen is electron rich and both carbon and hydrogen are electrophilic, as shown in the figure below. The electronegativity of oxygen is substantially greater than that of carbon and hydrogen. Unlike the alkyl halides, this group has two reactive covalent bonds, the C–O bond and the O–H bond.

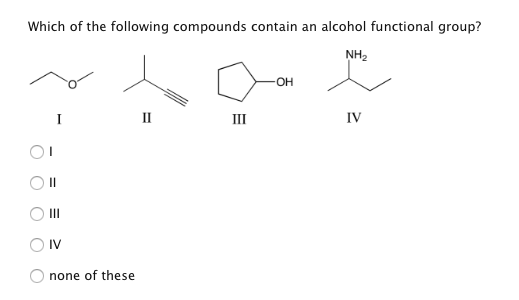
The functional group of an alcohol is the hydroxyl group, –OH. Alcohols contain the hydroxy functional group (-OH), bonded to a carbon atom of an alkyl or substituted alkyl group. They can be prepared from and converted into many different types of compounds.
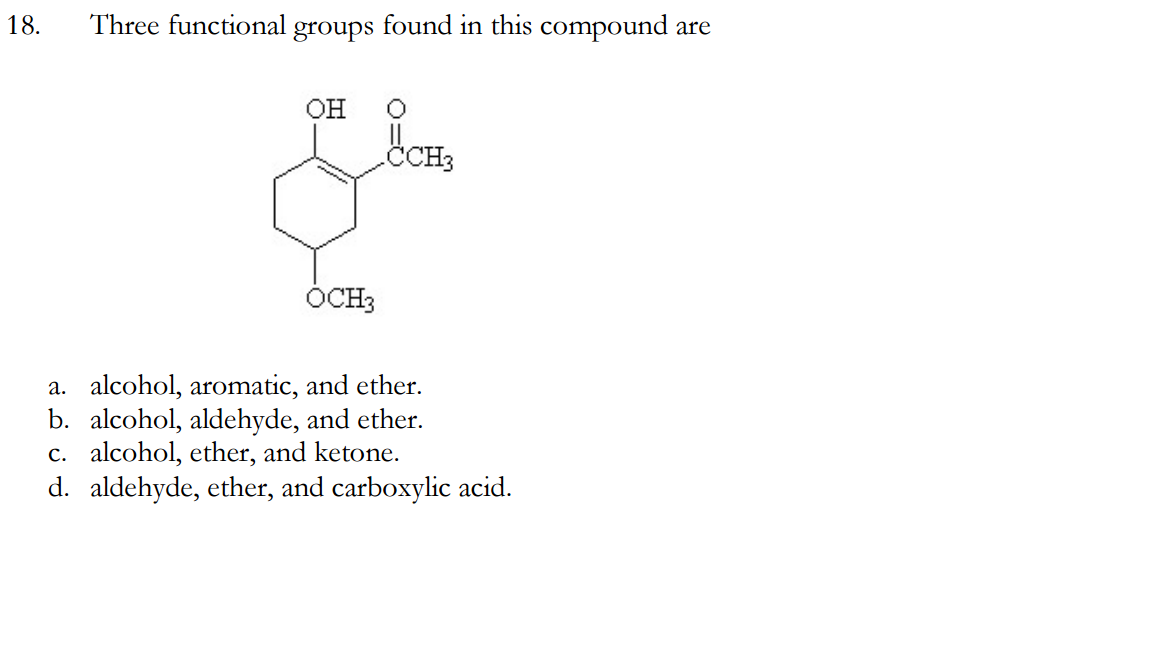
\( \newcommand\)Īlcohols are some of the most important molecules in organic chemistry.
